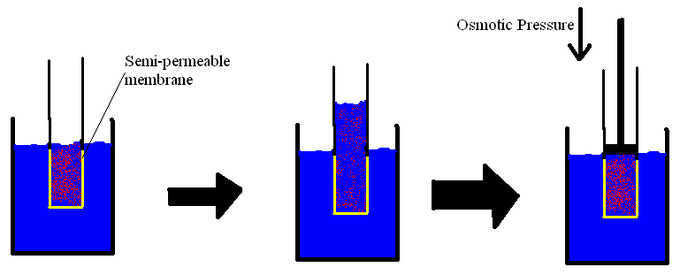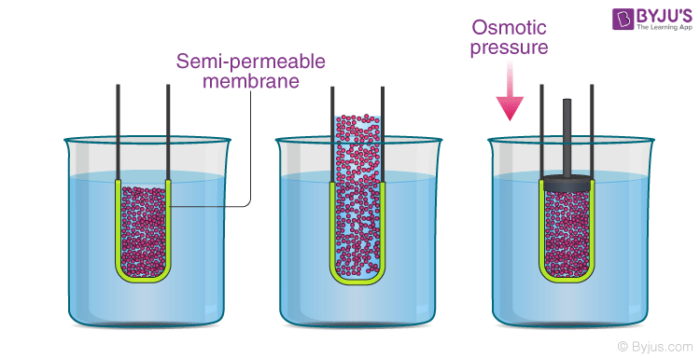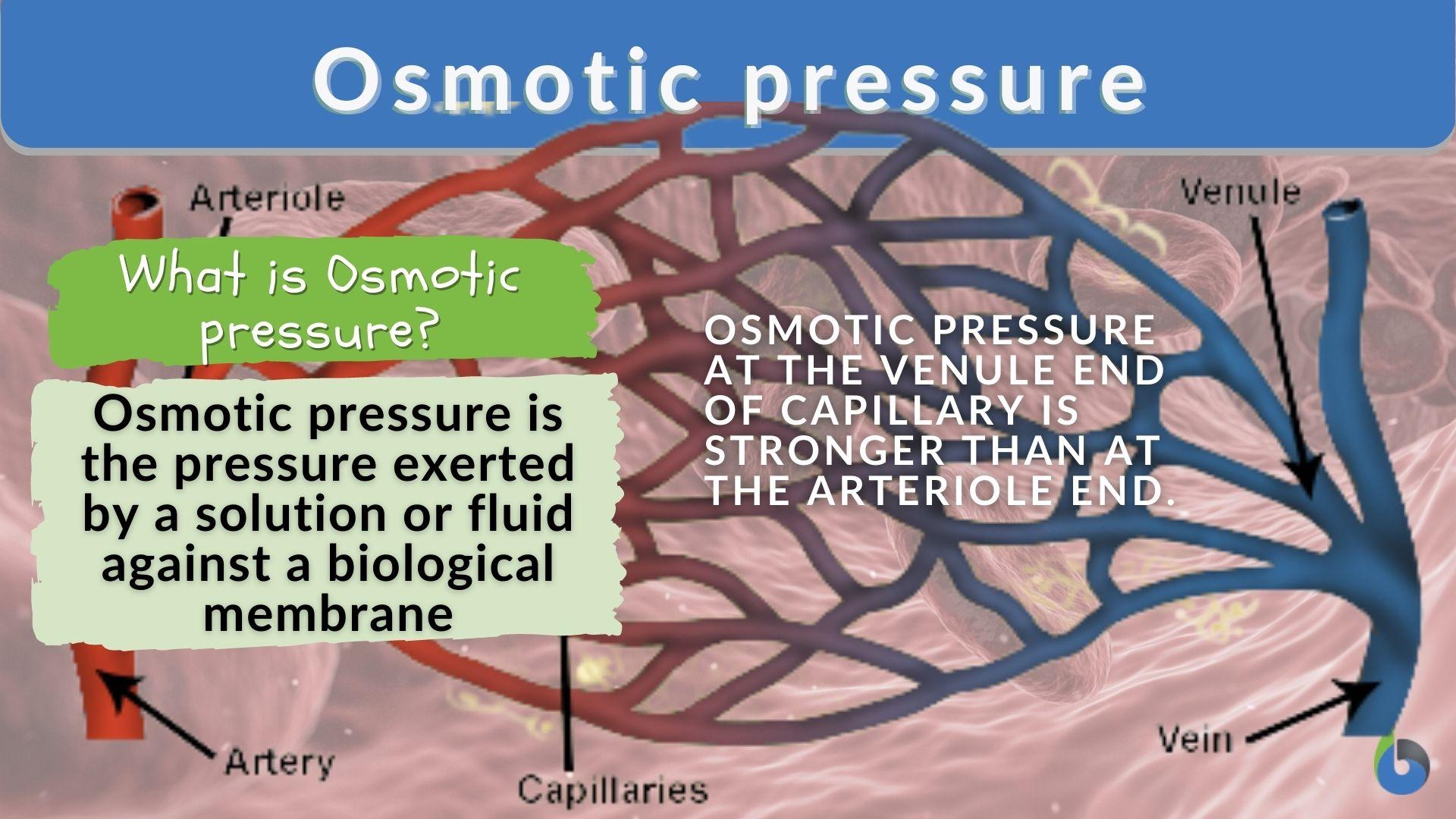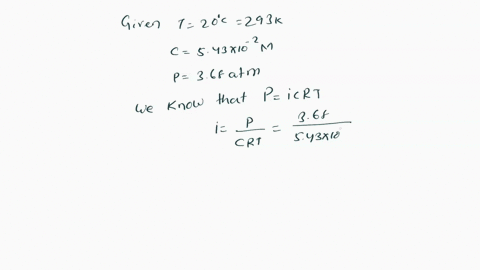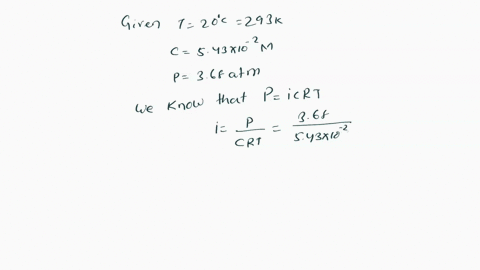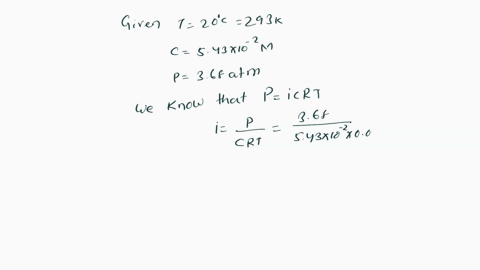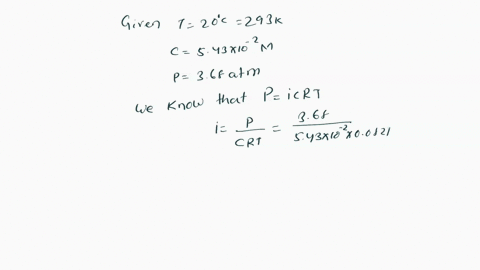
SOLVED: Π = - iMRT Where, Π is osmotic potential (or pressure), usually expressed in Megapascal (MPa) I is van 't Hoff factor This factor corrects for the dissociation of ionic compounds

1. An illustration of the ICRT construction with four point process {P... | Download Scientific Diagram

CHEM106S16 E3.pdf - CHEM 106 E3 Spring 2016 Name Constants and equations: 1 J = 1 kg∙m2∙s-2 Π=iCRT ΔT=iKfpm ΔT=iKbpm C=kP | Course Hero

Amazon.com : Personalized Custom 1 Line Noah's Ark with Rainbow Low Profile Thin Mouse Pad Mousepad : Office Products

A mixture of He and SO2 at one bar pressure contains 20% by weight of He . Partial pressure of He be:
Q 17/30 At 12^° C the osmotic pressure of a urea solution is 500mm . The solution is diluted and the temperature is raised to 27^° C, when the osmotic pressure is
![Welcome to Chem Zipper.com......: Decimolar solution of K4[Fe(CN)6] dissociate by 60% at 27°C. Determine osmotlc pressure in Nature/M2. Welcome to Chem Zipper.com......: Decimolar solution of K4[Fe(CN)6] dissociate by 60% at 27°C. Determine osmotlc pressure in Nature/M2.](https://lh3.googleusercontent.com/-Scet3fcHeWw/XsiaHk8xMpI/AAAAAAAAHm8/gwhzoNws9YYtSmNesnNkgkSgyki5Fg2aACLcBGAsYHQ/s1600/1590204946192896-0.png)
Welcome to Chem Zipper.com......: Decimolar solution of K4[Fe(CN)6] dissociate by 60% at 27°C. Determine osmotlc pressure in Nature/M2.

SOLVED: Π = - iMRT Where, Π is osmotic potential (or pressure), usually expressed in Megapascal (MPa) I is van 't Hoff factor This factor corrects for the dissociation of ionic compounds

The avrage osmotic presure of human of blood is 7.8 bar at 27^(@)C. The concentration of an aqueous NaCI solution that could be used in the blood stream is :